


50 years on: how inhaled
corticosteroids have changed
the treatment of asthma
Fernando M de Benedictis
, et al. LANCET RESPIR MED
2026 May 7:S2213-2600(26)00082-2.
Search strategy and selection criteria
•We searched MEDLINE, Current Contents, PubMed, and the Cochrane Library for articles published in English between Jan 1, 1972, and Sept 30, 2025, using the keywords “asthma”, “inhaled steroids”, “corticosteroids”, “recurrent wheeze”, “children”, and “adults”. We included original articles, reviews, randomised controlled trials, and other types of clinical research that focused on children 18 years and younger, and adults. We largely selected publications published between 1990 and 2025. We cite review articles to provide readers with additional details and more references than this Personal View can include. We also consulted our personal archives of references.
ABSTRACT
•Inhaled corticosteroids are the foundation of asthma therapy and now, 50 years on from their introduction, is an appropriate time to summarise some of the key studies that have progressed the field. We can now make better decisions in selecting the optimal inhaled corticosteroid-based regimens and identifying likely responders, based on biomarkers and patient characteristics. Inhaled corticosteroids reduce the risk of asthma attacks, but do not alter the course of the disease. Asthma remission, which is as yet an undefined therapeutic goal, is the aim, but the role of inhaled corticosteroids is unclear. High-dose inhaled corticosteroid therapy can cause systemic adverse events, suggesting that steps be taken to avoid this through the addition of long-acting β2-adrenergic agonists and the judicious use of biologics. Researchers will continue to learn more about the advantages and limitations of inhaled corticosteroids as they explore methods of disease prevention and remission in the future with new tools and treatments.
Key messages
•Inhaled corticosteroids are the mainstay of asthma management
•Inhaled corticosteroids do not prevent the development of asthma and do not cure asthma
•Most of the benefit in the long-term treatment of asthma is obtained with low-to-medium doses of inhaled corticosteroids
•Biomarkers and patient characteristics can help decision making in selecting the optimal inhaled corticosteroid-based regimen
•When asthma is uncontrolled by low-dose inhaled corticosteroids, adding a long-acting β2-agonists is the preferable step-up therapy in school age children, adolescents, and adults
•The effect of short courses of high-dose inhaled corticosteroids in managing asthma deterioration is uncertain
•As-needed, combination inhaled corticosteroid– formoterol inhaler is now considered the preferred reliever therapy in adults and adolescents 12 years and older
•Inhaled corticosteroids reduce the risk of exacerbations of asthma during pregnancy
•Regular use of inhaled corticosteroids might induce adverse events, especially for medium-to-high doses
•Insufficient availability and underuse of inhaled corticosteroids are key issues in low-income and middle-income countries
INTRODUCTION
•First oral and then inhaled corticosteroids were brought into clinical use by Harry Morrow-Brown, who showed they were effective in asthmatic adults with sputum eosinophilia.1 The introduction of inhaled corticosteroids as anti-inflammatory drugs for asthma management, initially in adults and then in children,2 revolutionised the approach to this disease, which historically had been based on the exclusive use of non-selective bronchodilators, with associated risks.3 In the 1980s, fundamental studies were conducted by John Toogood who set the stage for inhaled corticosteroid use in adults.4 However, in the therapeutic excitement of these results, Morrow-Brown’s lessons of personalised medicine were forgotten and researchers and clinicians fell headlong into the trap of thinking if some inhaled corticosteroid is good, more is better, uncritically escalating doses to the point of risking major side-effects for no additional benefit, and forgetting that not everyone who wheezes will benefit from them.
•50 years on from the seminal observation that sputum eosinophilia predicts inhaled corticosteroid response, researchers are now moving from umbrella terms, such as asthma, to a treatable trait approach.5 Although not everyone agrees with this approach, treating eosinophilic airway disease with an anti-eosinophil strategy is logical. The converse, using the same strategy when there is no sputum eosinophilia, was exactly what Morrow-Brown rejected. Whatever the airway disease, the first step is deconstructing the airway to identify treatable traits such as eosinophilic airway inflammation, bronchodilator reversible airway obstruction, and airway infection.6 When this identification is done, it becomes clear that some patients with the label asthma have no airway eosinophilia, and some patients with the labels chronic obstructive pulmonary disease (COPD) and bronchiectasis (conventionally thought of as neutrophilic diseases) also have eosinophilic airway inflammation and could benefit from inhaled corticosteroids. This treatable trait approach can enable identification of other overlapping disorders that can present with symptoms that might be indistinguishable from asthma, comorbidities that might require specific treatment, and environmental factors that, if addressed, might lead to better control of asthma rather than simply increasing inhaled corticosteroid treatment.7
•For true intellectual rigour, the treatable trait approach should be applied to all patients presenting with possible asthma. Guidelines have stressed the need for objective diagnosis in all cases of asthma;8 however, in many settings, particularly primary care, this might not be feasible, and it could be argued that a patient with typical features of the disease and a good response to low-dose inhaled corticosteroids does not require further testing.
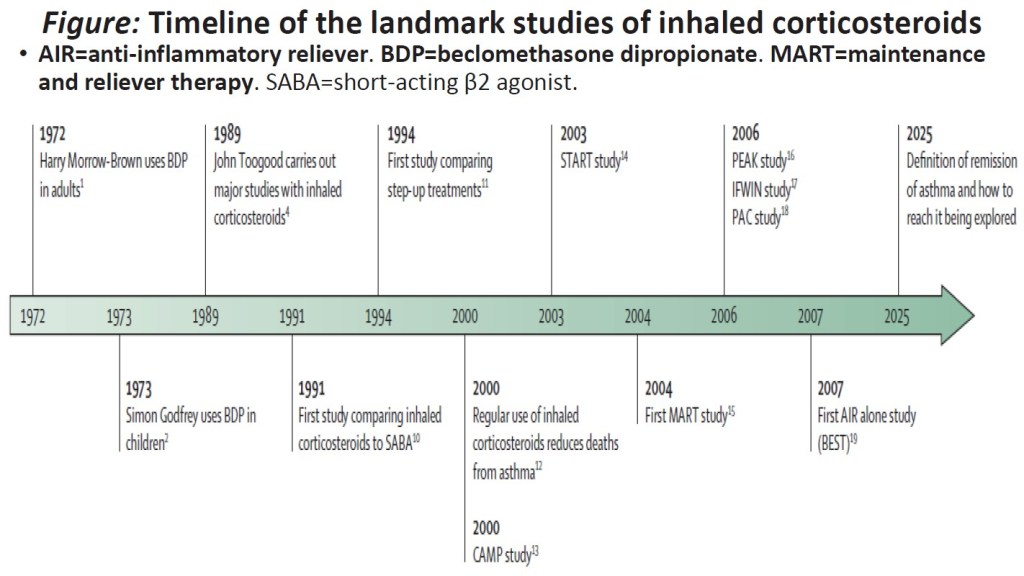
•Existing knowledge on mechanism of action, pharmacokinetics, pharmacodynamics and methods of administration of inhaled corticosteroids has been extensively reviewed elsewhere9 and will not be discussed in this Personal View. The aim of this Personal View is to discuss the evolving roles of inhaled corticosteroids in the treatment of asthma in the 50 years since their introduction into the clinic. We focus on pivotal studies that have provided unique perspectives but do not comprehensively review all inhaled corticosteroid studies. A timeline of the landmark studies is shown in the figure.
Figure: Timeline of the landmark studies of inhaled corticosteroids
•AIR=anti-inflammatory reliever. BDP=beclomethasone dipropionate. MART=maintenance and reliever therapy. SABA=short-acting β2 agonist.
Clinical efficacy
The effect of inhaled corticosteroids on asthma mortality
•Inhaled corticosteroids are the most effective anti-inflammatory medication for controlling asthma long-term.20 Importantly, after inhaled corticosteroids were introduced into clinical practice there was a rapid and substantial decrease in asthma mortality. A case-controlled study involving 30 569 patients with asthma showed that the regular use of inhaled corticosteroids was associated with a dose-dependent decreased risk of death from asthma, and that their discontinuation could be detrimental.12 In many low-income and middle-income countries (LMICs), inhaled corticosteroids are still not accessible to patients with asthma. Making them widely available in LMICs is also dramatically effective in reducing morbidity and mortality.21
The role of inhaled corticosteroids in improving asthma control and reducing exacerbations
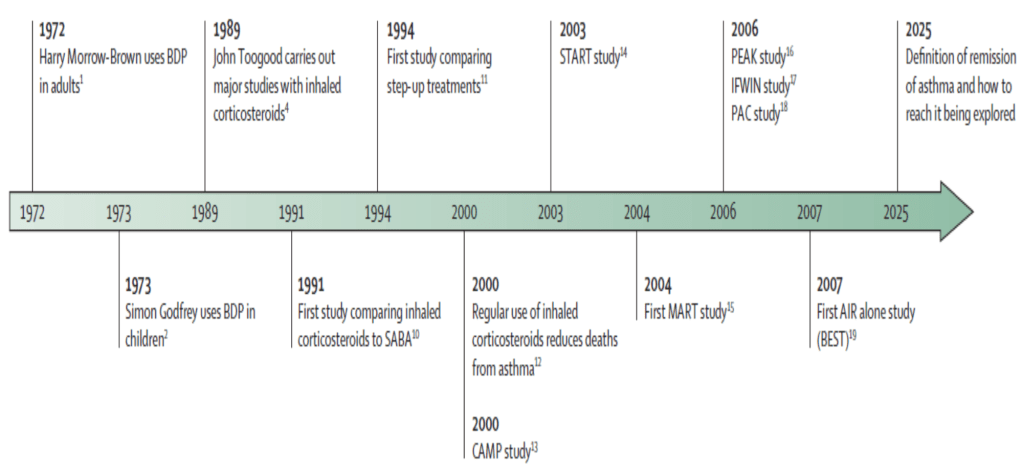
•The role of regular therapy with inhaled corticosteroids in improving asthma has been documented in many large studies in adults and children. The Childhood Asthma Management Program (CAMP) evaluated 1041 children aged 5–12 years with mild-to-moderate asthma randomised to receive budesonide, nedocromil, or placebo for a mean period of 4·3 years. When compared with children assigned to placebo, those given budesonide had fewer hospitalisations and urgent health-care visits, less albuterol use, and fewer courses of prednisone.13 The 3-year inhaled Steroid Treatment As Regular Therapy (START) study, including 7165 patients aged 5–66 years with recent onset mild persistent asthma, showed that low-dose budesonide decreased the risk of severe exacerbations and improved asthma control.14 During the subsequent 2-year open-label treatment with budesonide, patients who had received budesonide during the double-blind phase used significantly less additional asthma medication and had a lower risk of severe asthma-related events than those who had received placebo (table 1).22 In a Finnish study, adults with mild asthma who received inhaled corticosteroids within 12 months of asthma onset or after a 2-year delay showed no difference in clinical and functional variables after a 10-year follow-up; however, the delayed therapy group exhibited slightly worse disease control and airway inflammation.23 These data support the clinical benefit of early versus late intervention with inhaled corticosteroids in patients with asthma.
The effects of inhaled corticosteroids on lung function
•At the end of the treatment period of the CAMP study, the change in the forced expiratory volume in one second (FEV1) before, but not after, the administration of a short-acting β2-agonist (SABA) was significantly higher, and airway responsiveness to methacholine was significantly improved in children receiving budesonide, as compared with placebo.13 Over the 3-year double-blind phase of the START study, budesonide significantly increased both pre-bronchodilator and post-bronchodilator FEV1 compared with placebo.14 These results appeared to be mediated, at least in part, through an effect on reducing asthma exacerbations. In a post-hoc analysis, severe asthma exacerbations were associated with a more rapid decline in lung function, which was attenuated by budesonide.24 Another post-hoc analysis showed that there was no differential response to budesonide versus placebo for any clinical and functional outcome across different subgroups defined by symptom frequency (table 1).25
Table 1: Summary of some of the main long-term studies evaluating the efficacy of inhaled corticosteroids in asthma
•FEV1=forced expiratory volume in 1 s. NR=not reported.
The interpatient variability of response
•After the administration of inhaled corticosteroids, symptom control and improvement in lung function occur within the first weeks, with maximum improvement usually occurring within 1–2 months.26 Longer treatment and sometimes higher doses might be required to improve airway hyper-responsiveness (AHR).27
•Several studies reported interpatient variability in response to inhaled corticosteroids. In a multicentre trial, good (>15%) FEV1 response was associated with higher fractional exhaled nitric oxide (FeNO) levels compared with a poor (<5%) response (median 17·6 vs 11·1 ppb), suggestive of type-2 inflammation, high bronchodilator reversibility (25·2% vs 8·8%), and low FEV1–forced vital capacity (FVC) ratio (0·63 vs 0·73) before treatment. When compared with poor (<1 doubling dilution) improvement, excellent (>3 doubling dilutions) improvement in methacholine provocation concentration, resulting in a 20% decrease in FEV1 (PC20), was associated with higher sputum eosinophil levels (3·4% vs 0·1%) and older age at onset of asthma (20–29 years vs <10 years).28 In an 18-week study in children aged 6–17 years with mild-to-moderate asthma, response to fluticasone in terms of improvement in FEV1 was associated with higher levels of FeNO, total eosinophil count, and serum total IgE and lower levels of pulmonary function and methacholine PC20 compared with non-responders.29
•The Predicting Response to Inhaled Corticosteroid Efficacy (PRICE) study evaluated potential biomarkers predicting the response to short-term (6-week) inhaled corticosteroid therapy followed by a double-blind randomised controlled trial (RCT) of 16 additional weeks.30 Strong correlations with inhaled corticosteroid treatment success were low percentage predicted FEV1 and FEV1–FVC ratio and bronchodilator reversibility. When responders and non-responders were classified according to FEV1 improvement (>5% or ≤ 5% change from baseline), the short-term responders showed longer term asthma control only if maintained on inhaled corticosteroids. For the non-responders, asthma control remained unchanged whether inhaled corticosteroids were continued or substituted with placebo. Therefore, a short-term inhaled corticosteroid trial based on FEV1 change might be a good indicator of longer term asthma control in patients who are able to perform lung function testing, if FeNO and blood eosinophil counts are not available.
Comparison of inhaled corticosteroids with other anti-asthmatic drugs
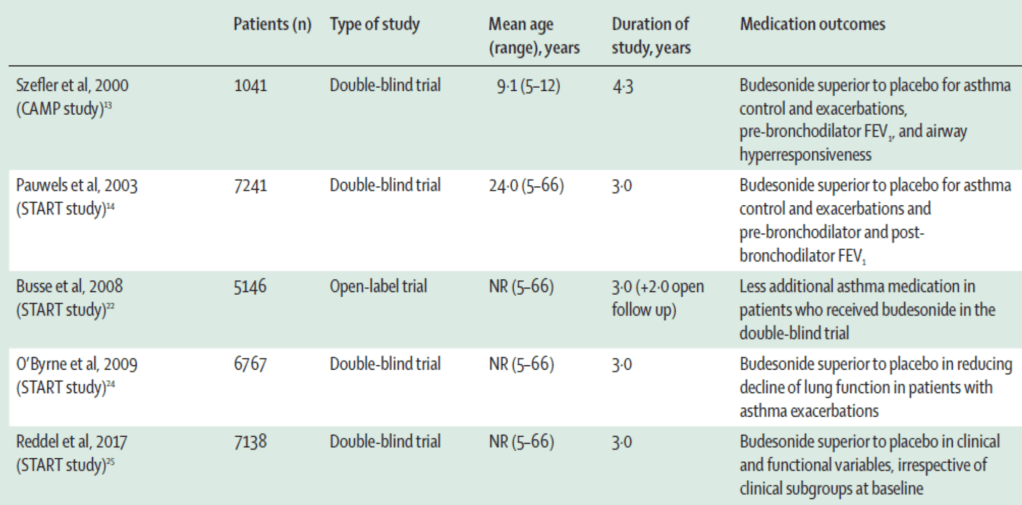
•When compared with regular treatment with SABA,10,31 salmeterol,32 nedocromil,13 or montelukast,29,33 daily treatment with inhaled corticosteroids resulted in better improvement of symptoms, lung function, and AHR, and reduction in number and severity of exacerbations. In addition to a better asthma control and improved lung function, inhaled corticosteroids were superior to montelukast at reducing inflammatory biomarkers in asthmatic children,34,35 adolescents, and adults.36 In the Pediatric Asthma Controller Trial (PACT) for children age 6–14 years with mild-to-moderate asthma fluticasone was superior to montelukast at modifying the exacerbation risk;37 parental history of asthma, history of inhaled corticosteroid use, low methacholine PC20, and high FeNO levels predicted the best long-term outcomes with fluticasone compared with montelukast (table 2).38 Regular administration of inhaled corticosteroids also reduced exercise-induced bronchoconstriction more consistently than leukotriene receptor antagonists (LTRAs).39
Table 2: Summary of some of the main studies comparing inhaled corticosteroids to other asthma medications
•AHR=airway hyperresponsiveness. FeNO=fractional exhaled nitric oxide. FEV1=forced expiratory volume in 1 s. PEF=peak expiratory flow.
Comparison of inhaled corticosteroids with other anti-asthmatic drugs
•These studies suggested that asthma therapy might move to a more individual, phenotypic-based strategy. One potential barrier is that there is little biomarker-stratified information on the effect of inhaled corticosteroids on longer term asthma outcomes such as exacerbation frequency, lung growth in children, and decline in lung function in adults. In one study involving adults with mild asthma, regular inhaled corticosteroid treatment was superior to as needed salbutamol in preventing severe exacerbations in patients with blood eosinophils more than 300 cells per μL but showed a trend to be inferior to salbutamol in the subgroup of patients with blood eosinophils less than 150 cells per μL.40 There is also compelling evidence that inhaled corticosteroids are not effective against exacerbations and decline in FEV1 in patients with COPD who have a low eosinophil count.41 Finally, a meta-analysis including several controlled intervention studies suggested that inhaled corticosteroid treatment guided by objective measures of eosinophilic inflammation is superior to guideline-based, symptom-directed management.42 Collectively, this evidence supports a biomarker-directed approach to inhaled corticosteroid treatment in asthma.43
•Since small airway (<2 mm diameter) inflammation results in increased air trapping and AHR, and is associated with uncontrolled asthma,44 small-particle-generating (or ultrafine particle) inhaled corticosteroid metered dose inhalers might provide enhanced control because of improved delivery to the small airways.45 Despite initial promising results, subsequent trials have not shown improved outcomes for the ultrafine particle devices when administered in clinically similar doses.46 Improved methods of measuring small airway disease and large-scale trials targeted at patients with clinically significant small airway inflammation are required to determine if targeted therapy provides clinically relevant improvement in asthma control.
Do inhaled corticosteroids prevent the development of asthma?
•Since inhaled corticosteroids are an excellent treatment for control of asthma, it was supposed that early treatment with these drugs would prevent the development of the disease. Three RCTs in infants with recurrent wheeze, two using continuous16,17 and one intermittent inhaled corticosteroid just at the time of viral wheeze,18 showed conclusively that this approach did not work. Factors initiating eosinophilic asthma and those that propagated the disease appear to be different.
The effect of inhaled corticosteroids on the course of asthma
•Inhaled corticosteroids do not cure asthma and do not alter the natural progression of the disease. When they are discontinued, deterioration of clinical control, lung function, and AHR follows within weeks in most adults47 and children.13,16,48
•Patients with asthma might lose lung function more rapidly than controls.49,50 A large cohort study showed that asthma can result in lung function abnormalities that increases in magnitude from age 5–18 years compared with controls, irrespective of treatment with inhaled corticosteroids.51 In the CAMP study, children who had received budesonide did not differ from those given placebo in terms of lung function at the end of 4·8-year follow-up period, during which the treatment was managed by the participants’ physicians.52 When the CAMP cohort was evaluated aged 20–30, four trajectories in the growth and decline in FEV1 were found: typical pattern (~25%), typical growth and early decline, reduced growth, and reduced growth and early decline, which were unrelated to treatment allocation.53 In the START study, the significant differences in pre-bronchodilator and post-bronchodilator FEV1 between budesonide and placebo groups during the double-blind phase were no longer significant at the end of the open-label budesonide phase due to improvement of lung function in the group that had previously received placebo.22
•In a meta-analysis of 13 RCTs and 11 observational studies, inhaled corticosteroid use was associated with modest improvement of pre-bronchodilator FEV1 across all age groups, the greatest benefits being observed in the first year of treatment; improvements in post-bronchodilator FEV1 were observed in adults but not in children.54 Such age-related differences are difficult to explain, but lung function trajectory studies have shown that small deficits in spirometry established early in life can diminish with time, some children with below-typical lung function entering a catch-up phase during puberty and early adulthood.55,56 Potential reasons for the inability of inhaled corticosteroids to prevent reduction in lung function could be related to involvement of the hard to reach small airways or the presence of a corticosteroid-independent mechanisms such as neurogenic inflammation,57 subepithelial fibrosis, and increased deposition of elastin.58 It is likely that airway inflammation and structural airway wall changes are parallel processes, rather than inflammation causing remodelling. There is currently insufficient evidence to determine whether treatment with inhaled corticosteroids reduces the incidence of fixed airflow obstruction in later life.
The dose–response effect
•Systematic reviews comparing dose-response effects of inhaled corticosteroids in patients with asthma of different severity showed that most of the benefit in mild-to-moderate severity disease is from low-to-medium doses.59 A systematic review and meta-analysis of 14 RCTs that compared moderate with low beclomethasone-equivalent doses of inhaled corticosteroids in children age 3–18 years with persistent asthma revealed a small but statistically significant increase of moderate over low doses in improving FEV1, but there was no significant difference in terms of other outcomes.60 Factors associated with a diminished response to inhaled corticosteroids include genetic polymorphisms, smoking, severe asthma, obesity, and vitamin D insufficiency.61 Pharmacokinetic and pharmacodynamic studies showed that even low doses of most inhaled corticosteroids can generate high glucocorticoid receptor occupancy (>90%) across the entire dose interval at steady state, which might explain observations that most clinical benefit typically occurs at low doses (appendix p 1).62
•Beasley and colleagues emphasised that the traditional terminology of low, medium, and high doses of inhaled corticosteroids is not evidence based, and that prescribing based on this terminology could lead to the use of excessive doses.63 They also proposed a reclassification of inhaled corticosteroid doses in adults based on a standard daily dose, which is defined as 200–250 μg of fluticasone propionate or equivalent, representing the dose at which approximately 80–90% of the maximum achievable therapeutic benefit of inhaled corticosteroid is obtained in adult asthma across the spectrum of severity.63 The same approach should be adopted in children with the daily dose no higher than fluticasone propionate 200 μg or equivalent, based on the Best Add-on Therapy Giving Effective Responses (BADGER) study.64
•Asthma can be controlled with low doses of inhaled corticosteroids in most patients. However, observational data suggest that many patients are stepped up in much higher doses. Doses should never be stepped up uncritically without reviewing why standard doses are not working, for example due to wrong diagnosis, comorbidities, or poor adherence to treatment.65 Before the introduction of biologics, around 1% of patients needed regular treatment with oral corticosteroids. True steroid-resistant asthma occurs but is rare, and the mechanisms have been reviewed elsewhere.66
Comparison between different inhaled corticosteroids
•Therapeutic effects of inhaled corticosteroids are mainly related to the entity of lung deposition, which is largely dependent on the delivery device and drug particle size, and the intrinsic potency of the molecule (ie, glucocorticoid receptor affinity). Due to many confounding factors, a comparison between different inhaled corticosteroids is a complex challenge. The Asthma Clinical Research Network determined the doses producing similar systemic effects, that is, the microgram dose at which each inhaled corticosteroid produces an equivalent degree of cortisol suppression, for six inhaled corticosteroids.67 It should be emphasised that tables classifying total daily doses of different inhaled corticosteroids as low, medium, and high for practical use are based on product information and do not imply equipotency. Therefore, switching from one inhaled corticosteroid to another, or from one delivery device to another, might result in a decrease or increase in drug potency, thus the patient might be exposed to less asthma control or increased risk of adverse events.
Step-up treatment
•In a pilot study, adding salmeterol to inhaled corticosteroids was the preferable strategy when compared with doubling the inhaled corticosteroid dose in asthmatic adults uncontrolled by 200 μg twice daily inhaled beclomethasone.11 In the Formoterol And Corticosteroids Establishing Therapy (FACET) study in adults with persistent symptoms of asthma despite treatment with inhaled corticosteroid, the addition of formoterol was superior to a four-fold increased dose of inhaled corticosteroids (budesonide 200–800 μg per day) for asthma symptoms and lung function, but inferior for severe exacerbations, and patients treated with formoterol and the higher dose of budesonide had the greatest reduction in severe exacerbations.68 In a study of children age 6–16 years with moderate asthma, no additional benefit for any outcome was found when adding either salmeterol or more beclomethasone to a daily dose of 400 μg beclomethasone.69 In the Oxis and Pulmicort Turbuhaler In the Management of Asthma (OPTIMA) study in adolescents and adults with mild persistent asthma, adding formoterol to low-dose inhaled corticosteroids was more effective in reducing exacerbations and improving asthma control than doubling the inhaled corticosteroid dose.70
•A multicenter, double-blind study showed no difference between salmeterol–fluticasone when compared with double dose fluticasone in school-age children with symptomatic asthma despite regular treatment with low-dose fluticasone.71 In the BAGDER study in school age children with asthma uncontrolled by 100 μg twice daily fluticasone, adding long-acting β2-agonist (LABA) was significantly more likely to provide the best response compared with high-dose fluticasone or montelukast step-up.64 A step-up therapy trial in Black patients with poorly controlled asthma showed a better response to adding salmeterol than to increasing fluticasone in adults and adolescents, and children responded equally to both (appendix p 2).72 In a meta-analysis of nine studies in school-age children and adolescents with uncontrolled asthma, patients receiving LABA plus inhaled corticosteroids showed a decreased risk of asthma exacerbations compared with higher inhaled corticosteroid doses.73
•More data are needed on the effects of LABA plus inhaled corticosteroids in preschool (≤6 years) children.74 In a systematic review and meta-analysis of studies in adolescents and adults whose asthma was not controlled on medium-dose inhaled corticosteroids, adding LABA to inhaled corticosteroids reduced moderate-to-severe exacerbations and increased asthma control.75 The effect of adding a LABA on reducing exacerbations might be explained by a pharmacodynamic interaction allowing the combination to activate the glucocorticoid receptor at lower inhaled corticosteroid doses.76
•There were initial concerns about an increased risk of life-threatening exacerbations and asthma mortality with LABA,77 but meta-analyses78 and further trials in adults and adolescents79 and in children80 showed that, when given within the same inhaler with inhaled corticosteroids (so LABA could not be used as monotherapy), LABA were not associated with an increased risk of any adverse outcome.
•A meta-analysis comparing association of LTRA with inhaled corticosteroids with inhaled corticosteroids in the same or higher dose was limited by data available only in children 6 years and older. However, no difference in hospitalisations or need for oral steroids were observed between groups.81 A meta-analysis that compared LABA with LTRA as adjunctive therapy to inhaled corticosteroids supports the use of inhaled corticosteroids–LABA over additional LTRA.82 A meta-analysis in adults and adolescents showed that long-acting antimuscarinic antagonists (LAMAs) add-on therapy to inhaled corticosteroids was associated with a lower risk of asthma exacerbations, but benefit might not be greater than adding LABA to inhaled corticosteroids.83 In a systematic review in children age 6–11 years with symptomatic asthma despite inhaled corticosteroid therapy, adding tiotropium bromide was associated with significant improvements in FEV1 and modest increase of asthma control compared with placebo.84
•A systematic review and meta-analysis of 20 RCTs in children, adolescents, and adults showed that adding LAMA to inhaled corticosteroid–LABA (triple therapy) was significantly associated with fewer severe exacerbations and modest improvements in lung function and asthma control without significant differences in quality of life or mortality.85 The Clinical study in Asthma Patients receiving triple Therapy in a single Inhaler (CAPTAIN) study in patients with asthma uncontrolled despite inhaled corticosteroid–LABA showed that increasing from a medium to high inhaled corticosteroid dose was more effective at preventing severe exacerbations than an additional LAMA in those with high baseline FeNO levels and blood eosinophil count, but not in those with low biomarkers (appendix p 2).86 Hence treatment titration in asthma to high inhaled corticosteroid doses should be based on inflammatory status. Some triple therapy combinations are not licensed for asthma outside the USA and especially in Europe.
Role of inhaled corticosteroids in managing deterioration in asthma control
•In the Improving Asthma Control Trial (IMPACT), adding intermittent short-course open-label budesonide (800 μg twice daily) to regular budesonide (200 μg twice daily) or placebo in adults with mild asthma at worsening of asthma symptoms did not change the exacerbation rate.87 However, over a 1-year period, patients receiving daily budesonide therapy showed greater improvements in pre-bronchodilator FEV1, AHR, percentage of sputum eosinophils, FeNO, asthma control, and number of symptom-free days. A meta-analysis of eight studies comparing the clinical effectiveness of increased (usually double dose) versus stable doses of inhaled corticosteroids as part of a patient-initiated action plan for home management of exacerbations in patients with persistent asthma showed that increased inhaled corticosteroid dose does not change outcomes (prescribed oral corticosteroids and hospital admissions).88
•The effect of short courses of very high doses of inhaled corticosteroids in managing acute asthma attacks has been studied. In a pragmatic trial in 1992 adults and adolescents, a personalised self-management plan that included a temporary quadrupling of the dose at asthma deterioration resulted in fewer (hazard ratio 0·81) asthma exacerbations than in the control group.89 In a double-blind study in school-age children with mild-to-moderate asthma treated with daily inhaled corticosteroids, quintupling the dose at the early signs of asthma deterioration did not change outcomes and could be associated with diminished linear growth.90 These different results are difficult to reconcile. Increasing doses of inhaled corticosteroids might be effective only in patients with eosinophilic asthma or in those not receiving daily controller therapy. As with the chronic management of asthma, we need to move away from a one-size-fits-all approach to acute asthma attacks to personalised treatment.91,92
As-needed administration of inhaled corticosteroids with fast-acting β2-agonists
•For many years, conventional wisdom was to use inhaled corticosteroid–LABA twice daily, with SABA as reliever therapy. One highly undesirable effect was that it enabled patients to overdose on SABA while taking insufficient inhaled corticosteroids, both risk factors for asthma attacks including death.93 A new strategy, using a single inhaler (inhaled corticosteroid–formoterol) for maintenance and reliever therapy (MART) was shown to be safer and more effective.15 It also became apparent that intermittent use of inhaled corticosteroids could be effective when delivered at the same time as a fast-acting β2-agonist such as LABA formoterol and SABA albuterol. This led to the concept of anti-inflammatory reliever therapy as sole management of adults and children with mild asthma.
•The as-needed, combined administration of inhaled corticosteroid–formoterol is now considered the preferred reliever therapy in those 12 years and older, irrespective of asthma severity.8,94 This recommendation is based on a better compliance, ability to titrate the inhaled corticosteroid dose through the vehicle of bronchodilator use, the risks of SABA monotherapy, and the evidence that inhaled corticosteroid–formoterol reliever therapy markedly decreases the occurrence of severe asthma exacerbations compared with SABA reliever therapy, across the spectrum of asthma severity, when the same maintenance treatment is taken.95,96 Despite the potential benefits of inhaled corticosteroid– formoterol reliever therapy in adults and adolescents, insufficient evidence is currently available supporting their safety and efficacy in childhood.97 Data supporting the use of combination inhaled corticosteroid–albuterol reliever therapy in asthma are more scarce.
•In mild asthma, there is GRADE level 1 evidence of the greater efficacy and safety of combined inhaled corticosteroid–formoterol as needed in adults, adolescents, and children 5 years or older,98–102 and combined inhaled corticosteroid–albuterol as needed in adults,19,103 compared with SABA as needed alone. When the effect on lung function over 12 weeks was compared between combined inhaled corticosteroid–SABA and its single components, both budesonide and albuterol contributed to efficacy.104 In school-age children with mild asthma, the hazard ratios for asthma exacerbations were lower in the daily beclomethasone group associated with as-needed SABA than in the rescue beclomethasone plus SABA group.105 Reliever treatment with inhaled corticosteroids and either formoterol or albuterol, as combined19,98–103,106 or associated105,107 regimens, showed equal or less efficacy than regular inhaled corticosteroid treatment plus as-needed SABA in asthma symptom control, but this was below the minimal clinical important difference (table 3).
Table 3: Summary of some of the main studies comparing combined or associated reliever therapy with inhaled corticosteroids plus a fast-onset β2- agonist to a SABA alone
•Data for mean age is either range or age included in study. GINA=Global Initiative for Asthma. PEF=peak expiratory flow. SABA=short-acting β2-agonist.
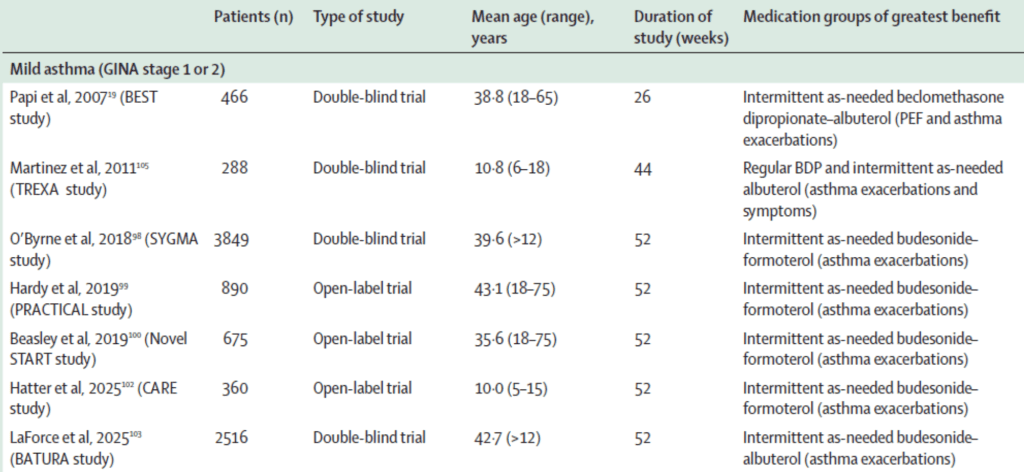
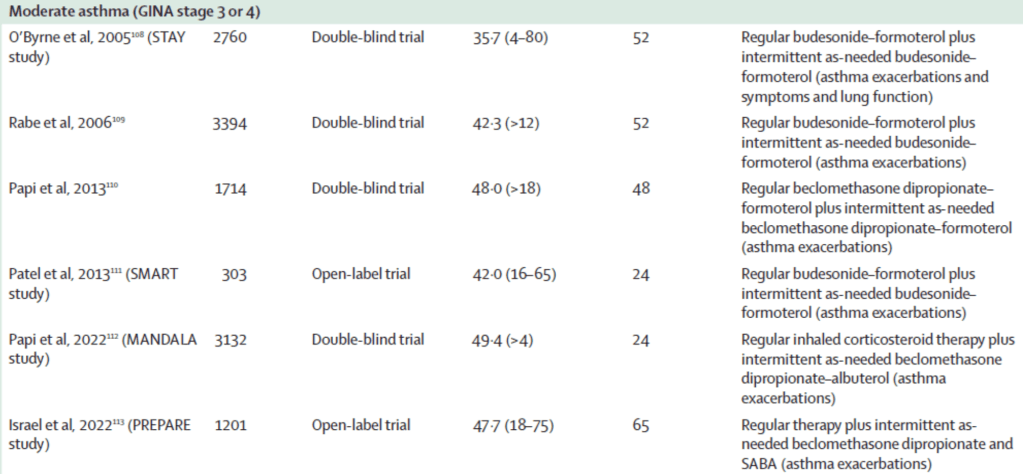
•In moderate asthma, there is level 1 evidence in adults, adolescents, and school-age children that combined inhaled corticosteroid–formoterol as reliever therapy reduces the risk of severe exacerbations compared with SABA reliever therapy, when the same baseline inhaled corticosteroid–LABA maintenance treatment is taken (MART regimen; table 3).108–111,114 The as-needed formoterol component also contributes to the reduction in exacerbation risk, due to its greater efficacy than as-needed SABA use.109 There is also evidence that inhaled corticosteroid–formoterol MART reduces the risk of severe asthma exacerbations compared with double the inhaled corticosteroid dose of maintenance inhaled corticosteroid–LABA and SABA reliever therapy.115
•This leads to a reduction in both oral and inhaled corticosteroid exposure and suggests that the timing of the inhaled corticosteroid–formoterol administration is important, in addition to the total daily inhaled corticosteroid dose, in terms of exacerbation risk reduction. Finally, inhaled corticosteroid–formoterol combination might also be used to prevent exercise-induced bronchoconstriction. When taken acutely before exercise, inhaled corticosteroid–formoterol provides greater protection than SABA; when taken long-term as a reliever and before exercise, inhaled corticosteroid– formoterol results in similar protection against exercise-induced bronchoconstriction as regular inhaled corticosteroid plus as-needed SABA.116 There is level 1 evidence that combined inhaled corticosteroid–albuterol as reliever therapy112 or use of an inhaled corticosteroid at the same time as albuterol reliever therapy113 reduces the risk of severe exacerbations compared with albuterol reliever alone in adults with uncontrolled asthma receiving a wide range of inhaled corticosteroid-containing maintenance therapies (table 3).
•A 2025 systematic review and network meta-analysis that included 27 RCTs (including 50 496 adults, adolescents, and school-age children) showed that compared with SABA reliever, inhaled corticosteroid– SABA was associated with a 16% reduction in risk of severe exacerbations and inhaled corticosteroid– formoterol reliever was associated with a 35% reduction, with no increase in adverse events; in the indirect comparison with inhaled corticosteroid–SABA, inhaled corticosteroid–formoterol was associated with a 22% reduction in risk of severe exacerbations.117 Compared with the use of as-needed inhaled corticosteroids and albuterol in separate inhalers, the use of inhaled corticosteroid–formoterol in a single inhaler as reliever therapy is cost-effective in patients 12 years or older with mild asthma.118
•Entrenched medical practice, cost, and regulatory complexities are major barriers to a widespread global transition to inhaled corticosteroid-containing reliever therapy. Furthermore, there are substantial gaps in research including a scarcity of studies of inhaled corticosteroid-containing reliever therapy in children.119
Inhaled corticosteroids in pregnancy
•Asthma symptoms can worsen, improve, or remain unchanged during pregnancy.120 Exacerbations are common, especially in the second trimester, and could be due to mechanical or hormonal changes, or to cessation or reduction of asthma medications due to concerns by the pregnant woman. Exacerbations and poor asthma control are associated with worse outcomes for both the baby (eg, pre-term delivery and low birthweight) and the pregnant woman (pre-eclampsia).121 Inhaled corticosteroids reduce the risk of exacerbation of asthma during pregnancy; they are very safe and adverse events in pregnancy have not been reported.122 Indeed, in a recent study, inhaled corticosteroids in pregnant women with asthma was associated with better lung function in their infants compared with those mothers who did not use inhaled corticosteroids in pregnancy.123 Pregnant women should be advised not to stop inhaled corticosteroid-containing medication, since cessation of medication is a risk factor for exacerbations.124 FeNO driven inhaled corticosteroid management of asthma in pregnancy has been shown to reduce asthma attacks,125 and the risk of perinatal complications and subsequent asthma in childhood.126,127
Inhaled corticosteroids in young children with recurrent wheeze
•Two meta-analyses of RCTs in toddlers with recurrent wheeze revealed benefit from regular or intermittent inhaled corticosteroids.128,129 An intermittent high-dose inhaled corticosteroid regimen in preschool children was as effective as a daily low dose with less total exposure to inhaled corticosteroids.130 Although intermittent administration of very high-dose inhaled corticosteroids in preschool children reduces the number of asthma attacks treated with prednisolone, adverse events can still occur.131
•All these studies adopted an inadequate one-size-fits-all approach.128–131 In the first attempt to personalise therapy in preschool children with asthma, three different treatments (daily fluticasone, intermittent fluticasone, and daily montelukast) were compared. Those with combined aeroallergen sensitisation and high blood eosinophil count had the best response to regular inhaled corticosteroid therapy; clear predictors of best response to intermittent inhaled corticosteroid therapy could not be identified.132
•In a post hoc analysis of data from three clinical trials of 1074 preschool children with recurrent wheezing, each blood eosinophil cut point was associated with higher exacerbation rate and more hospitalisations; the addition of atopic sensitisation was further associated with an improved response to daily inhaled corticosteroids.133 In future studies in preschool children with recurrent wheeze, patients should be stratified by eosinophil count and aeroallergen sensitisation to determine the effects of inhaled corticosteroids in this age group. Finally, it should be emphasised that treatment with inhaled corticosteroids in young children should only be initiated with the goal of producing a symptomatic benefit, such as reduction in exacerbations, as there is no evidence that early therapy prevents long-term progression to persistent asthma.
Adverse events of inhaled corticosteroids
•Although low-dose inhaled corticosteroid treatment is generally considered safe, there are potential adverse events of regular medium-to-high doses, both in adults134 and in children.135 Systemic absorption of inhaled corticosteroids might result from deposition in the oropharynx and airway, and swallowed medication that is not catabolised by hepatic first pass metabolism. Advances in formulation, the use of more sophisticated delivery devices, and attention to proper inhalation techniques have improved lung deposition of inhaled corticosteroids and concomitantly reduced local effects. Inhaled corticosteroid deposition in the airway is an inevitable source of systemic absorption.136 The development of new formulations with minimal systemic absorption that are specifically tested in preschool children should be encouraged.
•Adverse events are often underestimated in clinical practice, not least because some, such as adrenal suppression, might not be recognised clinically, and others, such as cataracts, osteoporosis, and diabetes, commonly occur in adults in whom their occurrence might be attributed solely to courses of oral corticosteroids taken over time or not considered related to their asthma treatment at all. Another concerning effect of inhaled corticosteroids in adults is the increased risk of tuberculosis,137 atypical Mycobacterial infection,138 and pneumonia.139,140 Since systemic steroids are immunosuppressive, it is not surprising that topical immunosuppression of airway immune functions might also be seen.141
•The effect of inhaled corticosteroids on growth in children has been a matter of concern. A meta-analysis of 16 RCTs showed that regular inhaled corticosteroid treatment significantly reduced growth velocity at 1-year follow-up (mean difference –0·48 cm per year).142 In the long-term follow-up of the CAMP study, mean adult height was 1·2 cm lower in the budesonide group than in the placebo group (p<0·001). Such decreased growth velocity occurred during the first 2 years of the study and was greatest in prepubertal participants.143
•It is also worth noting that absorption of inhaled corticosteroids is greater in healthy individuals and those with mild asthma compared with those with moderate-to-severe asthma,144 thus supporting the concept for step-down therapy in those who achieve asthma control. It should be emphasised that the benefits of inhaled corticosteroids, properly prescribed and used, far outweigh their potential adverse effects. Recommendations to mitigate the risk of adverse events in patients taking inhaled corticosteroids are shown in the panel.
Panel: Recommendations to mitigate the risk of adverse events in patients with asthma taking inhaled corticosteroids
•Be sure that the patient has asthma—the diagnosis of asthma needs always to be confirmed
•When asthma is confirmed, consider that most patients are controlled by low inhaled corticosteroid doses
•Consider as-needed inhaled corticosteroid therapy if asthma is intermittent
•Choose an appropriate delivery device to reach a high therapeutic index
•Adjust treatment on a regular basis by prescribing the minimal effective inhaled corticosteroid dose as overtreatment of asthma is common
•Before escalating treatment, check adherence and address modifiable environmental triggers
•If asthma is uncontrolled on low-dose inhaled corticosteroids, consider an add-on therapy rather than increasing the inhaled corticosteroid dose
•Check potential use of other steroids for concomitant conditions (eg allergic rhinitis or atopic dermatitis)
•In children, check growth at least every 6 months and plot on a growth chart
•Encourage exercise and optimise nutritional status
Availability and affordability of inhaled corticosteroids and inhaled corticosteroid–LABA in LMICs
•Although inhaled corticosteroids are widely available and used in high-income countries (HICs), minimal availability and underuse are key issues in LMICs. Data from the Global Asthma Network phase 1 across 25 countries found concerning insufficient use of inhaled corticosteroids among children with asthma even with severe symptoms, and corresponding overuse of SABA and oral bronchodilators, with poor asthma control. Inhaled corticosteroids were only used in 23% of children with severe asthma symptoms and inhaled corticosteroid– LABA were only used in 18% of LMICs.145
•A systematic review of availability and affordability of inhaled medicines for asthma in 60 LMICs found that inhaled therapy for asthma was only available in 30% of countries included.146 A cross-sectional survey of medication availability found that inhaled corticosteroids were available in 30% of pharmacies and 36% of hospitals, and combination inhaled corticosteroid–LABA was available in 11% of pharmacies and 5% of hospitals.147 Insufficient access to inhaled corticosteroids and cost, especially for inhaled corticosteroid–LABA, are key factors reducing use. Similarly, the cost of spacers could reduce the use of inhaled corticosteroids in children, but a low-cost spacer constructed from a plastic bottle is equally effective.148 Paradoxically, although overtreatment can occur in HICs, a scarcity of effective therapy means large numbers of adults and children receive suboptimal care and live with uncontrolled asthma. There is an urgent need for equitable access to affordable inhaled corticosteroids, inhaled corticosteroid–LABA, and spacers in all LMICs. Insufficient access is also a factor for populations in HICs who have no or inadequate health insurance.
Back to Morrow-Brown: from labels to cells
•The treatable trait approach has highlighted that some patients with what are thought traditionally as diseases of neutrophilic inflammation and sometimes infection, such as COPD and bronchiectasis, are also eosinophilic and respond to inhaled corticosteroids and even type-2 biologics. This finding underscores the importance of the treatable trait approach rather than umbrella labels such as asthma, COPD, and especially asthma-COPD overlap syndrome.149
The future: role of inhaled corticosteroids in the era of biologics
•Inhaled corticosteroids have come a long way since being reintroduced by Morrow-Brown, and his observation that eosinophils, not labels, predict response to inhaled corticosteroids has eventually been remembered. Even despite the advent of type-2 biologics, inhaled corticosteroids remain the foundation of treatment of eosinophilic airway disease. There is only scarce evidence that it is feasible to achieve an inhaled corticosteroid dose reduction in patients on biologics. Targeting eosinophils with benralizumab (anti-IL-5 receptor α) in patients with severe asthma has been shown to reduce the need for both systemic corticosteroids and high-dose inhaled corticosteroids (table 4).150–152 However, regular inhaled corticosteroids remain important in responders to benralizumab as a parallel reduction in FEV1 and increase in FeNO was seen when regular inhaled corticosteroid therapy was reduced to as-needed inhaled corticosteroids.150 Patients well-controlled on biologics are likely to want to reduce other treatments or are perhaps more likely to reduce them empirically.
Table 4: Studies that evaluated dose reduction of patients using inhaled corticosteroids with severe asthma controlled on biologics

•The key questions for the future are what asthma remission means (on treatment vs off treatment) and how it can be reached.153 It has been speculated that asthma remission, however it is defined, can be reached if biologics are introduced early.154–156 However, the current exorbitant costs mean that this strategy is unlikely to be achievable in the short-term. A detailed discussion of asthma remission, and whether this can be reached either by inhaled corticosteroids or biologicals, is beyond the scope of this Personal View. More likely, for the foreseeable future, we will attempt to control active asthma aggressively with inhaled corticosteroids, predominantly using inhaled corticosteroid–formoterol MART, and inhaled corticosteroids that will continue to play a major role in the treatment of all eosinophilic airway diseases.157